|
|
|
|
HOME NEWS RESEARCH PEOPLE PUBLICATIONS
PRESS TEACHING RESOURCES CONTACT |
|
|
RESEARCH Background Miniaturized devices have had a significant impact on our
society in revolutionizing the electronics industry. A similar trend of
size-reduction is emerging in the health care industry to produce small
bioanalytical devices for disease diagnosis, novel methods for drug delivery
to treat diseases, and microfabricated platforms for studying microorganisms.
Nanostructured materials, in particular, have revolutionized capabilities of
biotic-abiotic interfaces in various biomedical devices, including affinity
biosensors, implantable neural electrodes, drug delivery platforms, and in
vitro models. Despite the recent research on these materials, significant
challenges remain in controlling material properties, interfacing
nanocomponents with instrumentation, and engineering their interaction with
biological systems. Prior to joining UC Davis as a faculty member, Şeker’s
dissertation focused on microfabrication and thermo-mechanical
characterization of nanoporous gold [1,2], a promising candidate for
functional surface coatings due to its controllable morphology, electrical
conductivity, and well-studied gold surface chemistry [3]. In
addition, he studied mass-transport in porous media[4] and
synthesized a composite material that exhibits very low elastic modulus and
high electrical conductivity [5] –
highly desirable attributes for flexible electrodes. As a postdoctoral
researcher in chemistry department, his research extended to the study of
biomolecule-surface interactions [6] and
development of novel flow control methods in microfluidic circuits [7,8]. As a research associate at Center for
Engineering in Medicine (CEM), he began to utilize nanoporous metals for
developing planar multiple electrode arrays for electrophysiology
applications [9]. The overarching objective of our group is to utilize our
expertise at the intersection of nanoporous metal synthesis, microfluidics,
and device engineering to overcome challenges in the evolution of
miniaturized devices relevant to microelectronics and life sciences. We
gratefully acknowledge support from National Science Foundation (1512745, 1454426, 2003849),
UC Lab Fees Research Program (12-LR-237197),
National Institutes of Health (T32-GM008799, R21-EB024635,
R21-AT010933,
R03-NS118156,
R03-NS118156-S1,
R03-NS145146,
R01- EB034279), United States Department of Agriculture (NIFA
AFRI 2024-67017-42816), UC Davis College of Engineering Next Level
Research Funds, UC Davis Research Investments in the Sciences and Engineering
(RISE),
UC Davis Alzheimer’s Disease Research Center, UC Davis Environmental Health
Science Center, UC Davis Comprehensive Cancer Center, UC Davis Microbiome
Special Research Program. The
projects listed below are a selected group of our current research thrusts,
which are also summarized in a short video.
Please contact Prof. Erkin Şeker for
more information. |
|
|
Main
Research Thrusts |
|
|
Nanostructured
Electrochemical Biosensors An integral component of a biosensor platform with an
electrochemical read-out is the sensor element that interfaces biomolecular
detection probes with instrumentation electronics. Sensor performance, on
both biomolecular and electrical levels, is strongly influenced by sensor
coating properties, such as morphology, surface chemistry, and metallurgy.
While nanostructured materials have shown significant promise in enhancing
the performance of these sensors, the underlying mechanisms for this
enhancement are not fully understood. Currently,
we are fabricating novel nanostructured electrodes to provide insight into
how nanoscale features enhance sensitive measurement and purification of
nucleic acids in complex biological samples [10-18]. The fundamental studies
will reveal a set of design rules for the development of nanostructured
electrochemical sensor elements and assist technological advancements in food
safety, water quality, and medical diagnostics. |
Tunable nanoporous
gold morphology |
|
Multifunctional
Biomedical Device Coatings There is a significant need for medical devices that can both
monitor and modulate physiological activity, while minimizing adverse tissue
response to an implant. This is particularly important in neurological
disorders, since neurons exhibit both electrical and chemical activity as a
part of their normal function and implanted neural interfaces typically
suffer from deteriorating device performance due to tissue-material
interactions. Currently,
we are creating multifunctional neural electrodes that incorporate tunable
drug delivery and topographical cues. Specifically, we focus on the
fundamentals of molecular release from nanoporous metals [19-22] and cell-surface
interactions as a function of nano-topography [23-25]. We expect that these
efforts will translate into advanced neural interfaces for closed-loop
control of neurological disorders such as epilepsy. |
Astrocyte adhering
onto porous surface |
|
Nanoporous
Metal Morphology Libraries In the studies described above, nanoporous metal attributes
(e.g., characteristic feature size, effective surface area, electrical
conductivity, and surface chemistry) translate into several key device
performance metrics: (i) limit of detection, sensitivity, and
biofouling-resilience of affinity biosensors; (ii) biocompatibility,
signal-to-noise ratio, and charge injection capacity of neural electrodes;
and (iii) loading capacity, release kinetics, and on demand release in drug
delivery platforms. Currently,
we are developing techniques to modulate pore morphology (via
electro-thermo-mechanical mechanisms) and surface chemistry with the goal of
creating on-chip libraries that display multiple material attributes [14,26-29]. We expect these libraries
to allow for high-throughput study of structure-property relationships in the
context of biomolecule-tissue-material interactions, as well as other
applications such as catalysis and energy storage. |
Freestanding
nanoporous gold beam array |
|
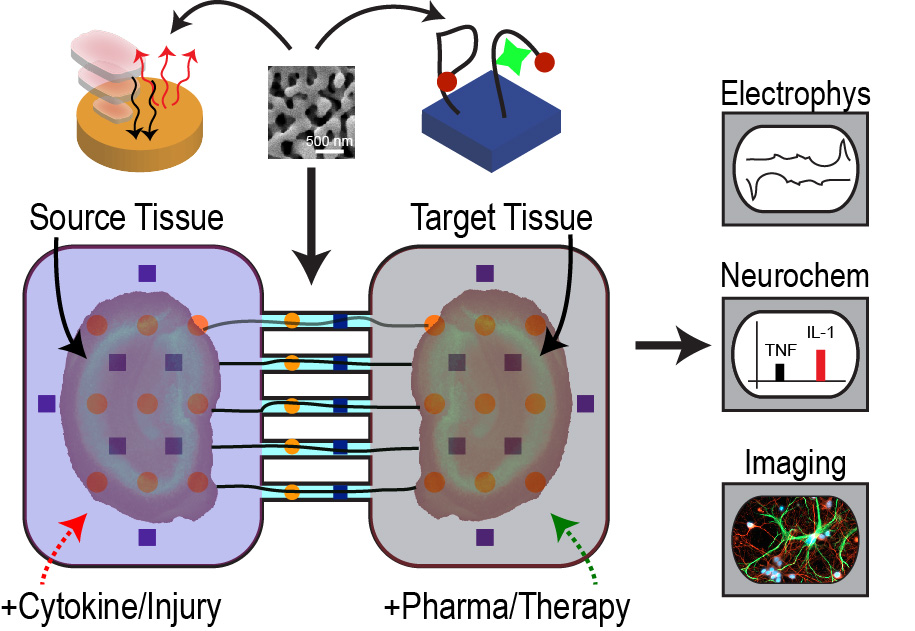
Microphysiological
Models Inflammation plays a key role in numerous conditions,
including degenerative diseases[30],
cancer, brain injury, Crohn's disease, and metabolic disorders. Gut
microbiota adds another level complexity to the picture, where bacterial
metabolites modulate inflammation and neural activity. There are still many
unknowns on the mechanisms of how inflammation traverse large anatomical
distances and how gut microbiota and the nervous system interact. Currently,
we are integrating our sensing and actuation technologies with microfluidics
and novel cell culture approaches to engineer microphysiological models of
neuroinflammation and enteric epithelium-neuron interaction [31-35]. These models, in turn,
allow us to study the transmission modes of inflammation in the central
nervous system and influence of bacterial metabolites. We expect these
platforms to facilitate rapid testing of therapeutics and provide insight
into unique physiological phenomena observed in vivo. |
Tissue chip with
multifunctional electrodes |
|
|
|
|
Reference Papers 1. Seker,
E., Gaskins, J., Bart-Smith, H., Zhu, J., Reed, M., Zangari, G., Kelly, R.
& Begley, M. The effects of post-fabrication annealing on the mechanical
properties of freestanding nanoporous gold structures. Acta Materialia 55:4593 (2007). 2. Seker, E., Reed, M. L. & Begley, M.
R. A thermal treatment approach to reduce microscale void formation in
blanket nanoporous gold films. Scripta
Materialia 60:435 (2009). 3. Seker, E., Reed, M. & Begley, M.
Nanoporous Gold: Fabrication, Characterization, and Applications. Materials 2:2188 (2009). 4. Seker, E., Begley, M., Reed, M. &
Utz, M. Kinetics of capillary wetting in nanoporous films in the presence of
surface evaporation. Applied Physics
Letters 92:013128 (2008). 5. Seker, E., Reed, M., Utz, M. &
Begley, M. Flexible and conductive bilayer membranes of nanoporous gold and
silicone: Synthesis and characterization. Applied
Physics Letters 92:154101 (2008). 6. Huang, L., Seker, E., Landers, J.,
Begley, M. & Utz, M. Molecular Interactions in Surface-Assembled
Monolayers of Short Double-Stranded DNA. Langmuir
26:11574 (2010). 7. Leslie, D., Easley, C., Seker, E.,
Karlinsey, J., Utz, M., Begley, M. & Landers, J. Frequency-specific flow
control in microfluidic circuits with passive elastomeric features. Nature Physics 5:231 (2009). 8. Seker, E., Leslie, D., Haj-Hariri, H.,
Landers, J., Utz, M. & Begley, M. Nonlinear pressure-flow relationships
for passive microfluidic valves. Lab on
a Chip 9:2691 (2009). 9. Seker, E., Berdichevsky, Y., Begley, M.,
Reed, M., Staley, K. & Yarmush, M. The fabrication of low-impedance
nanoporous gold multiple-electrode arrays for neural electrophysiology
studies. Nanotechnology 21:125504
(2010). 10. Daggumati, P., Matharu, Z. & Seker, E.
Effect of Nanoporous Gold Thin Film Morphology on Electrochemical DNA
Sensing. Analytical chemistry
87:8149 (2015). 11. Daggumati, P., Matharu, Z., Wang, L. &
Seker, E. Biofouling-resilient nanoporous gold electrodes for DNA sensing. Analytical chemistry 87:8618 (2015). 12. Daggumati, P., Appelt, S., Matharu, Z.,
Marco, M. & Seker, E. Sequence-Specific Electrical Purification of
Nucleic Acids with Nanoporous Gold Electrodes. Journal of the American Chemical Society 138:7711 (2016). 13. Zhou, J. C., Feller, B., Hinsberg, B.,
Sethi, G., Feldstein, P., Hihath, J., Seker, E., Marco, M., Knoesen, A. &
Miller, R. Immobilization-mediated reduction in melting temperatures of
DNA–DNA and DNA–RNA hybrids: Immobilized DNA probe hybridization studied by
SPR. Colloids and Surfaces A:
Physicochemical and Engineering Aspects 481:72 (2015). 14. Matharu, Z., Daggumati, P., Wang, L.,
Dorofeeva, T. S., Li, Z. & Seker, E. Nanoporous-Gold-Based Electrode
Morphology Libraries for Investigating Structure–Property Relationships in
Nucleic Acid Based Electrochemical Biosensors. ACS Applied Materials & Interfaces 9:12959 (2017). 15. Veselinovic, J., Alangari, M., Li, Y.,
Matharu, Z., Artés, J. M., Seker, E. & Hihath, J. Two-tiered electrical
detection, purification, and identification of nucleic acids in complex
media. Electrochimica Acta 313:116
(2019). 16. Veselinovic, J., Li, Z., Daggumati, P.
& Seker, E. Electrically Guided DNA Immobilization and Multiplexed DNA
Detection with Nanoporous Gold Electrodes. Nanomaterials 8:351 (2018). 17. Veselinovic, J., AlMashtoub, S., Nagella,
S. & Seker, E. Interplay of Effective Surface Area, Mass Transport, and
Electrochemical Features in Nanoporous Nucleic Acid Sensors. Analytical chemistry (2020). 18. Veselinovic, J., Almashtoub, S. &
Seker, E. Anomalous trends in nucleic acid-based electrochemical biosensors
with nanoporous gold electrodes. Analytical
chemistry 91:11923 (2019). 19. Seker, E., Berdichevsky, Y., Staley, K. J.
& Yarmush, M. L. Microfabrication-Compatible Nanoporous Gold Foams as
Biomaterials for Drug Delivery. Advanced
Healthcare Materials 1:172 (2012). 20. Kurtulus, O., Daggumati, P. & Seker,
E. Molecular Release from Patterned Nanoporous Gold Thin Films. Nanoscale 6:7062 (2014). 21. Polat, O. & Seker, E. Halide-Gated
Molecular Release from Nanoporous Gold Thin Films. The Journal of Physical Chemistry C 119:24812 (2015). 22. Li, Z., Polat, O. & Seker, E. Voltage‐Gated
Closed‐Loop Control of Small‐Molecule
Release from Alumina‐Coated Nanoporous Gold Thin Film Electrodes. Advanced Functional Materials:1801292
(2018). 23. Chapman, C. A., Chen, H., Stamou, M.,
Biener, J., Biener, M. M., Lein, P. J. & Seker, E. Nanoporous Gold as a
Neural Interface Coating: Effects of Topography, Surface Chemistry, and
Feature Size. ACS Applied Materials and
Interfaces 7:7093 (2015). 24. Chapman, C. A. R., Wang, L., Chen, H.,
Garrison, J., Lein, P. J. & Seker, E. Nanoporous Gold Biointerfaces:
Modifying Nanostructure to Control Neural Cell Coverage and Enhance
Electrophysiological Recording Performance. Advanced Functional Materials 27:1604631 (2017). 25. Hampe, A., Li, Z., Sethi, S., Lein, P.
& Seker, E. A Microfluidic Platform to Study Astrocyte Adhesion on
Nanoporous Gold Thin Films. Nanomaterials
8:452 (2018). 26. Chapman, C. A., Daggumati, P., Gott, S.
C., Rao, M. P. & Seker, E. Substrate topography guides pore morphology
evolution in nanoporous gold thin films. Scripta
Materialia 110:33 (2016). 27. Chapman, C. A., Wang, L., Biener, J.,
Seker, E., Biener, M. M. & Matthews, M. J. Engineering on-chip nanoporous
gold material libraries via precision photothermal treatment. Nanoscale 8:785 (2016). 28. Dorofeeva, T. S., Matharu, Z., Daggumati,
P. & Seker, E. Electrochemically Triggered Pore Expansion in Nanoporous
Gold Thin Films. The Journal of
Physical Chemistry C 120:4080 (2016). 29. Dorofeeva, T. S. & Seker, E. In Situ
Electrical Modulation and Monitoring of Nanoporous Gold Morphology. Nanoscale 8:19551 (2016). 30. Kim, H., Le, B., Goshi, N., Zhu, K.,
Grodzki, A. C., Lein, P. J., Zhao, M. & Seker, E. Primary cortical cell
tri-culture to study effects of amyloid-β on microglia function and
neuroinflammatory response. Journal of
Alzheimer’s Disease 102:730 (2024). 31. Goshi, N., Girardi, G., da Costa Souza,
F., Gardner, A., Lein, P. J. & Seker, E. Influence of microchannel
geometry on device performance and electrophysiological recording fidelity
during long-term studies of connected neural populations. Lab on a Chip 22:3961 (2022). 32. Goshi, N., Kim, H. & Seker, E. Primary
Cortical Cell Tri-Culture-Based Screening of Neuroinflammatory Response in
Toll-like Receptor Activation. Biomedicines
10:2122 (2022). 33. Goshi, N., Morgan, R. K., Lein, P. J.
& Seker, E. A primary neural cell culture model to study neuron,
astrocyte, and microglia interactions in neuroinflammation. Journal of neuroinflammation 17:1
(2020). 34. Kim, H., Girardi, G., Pickle, A., Kim, T.
S. & Seker, E. Microfluidic tools to model, monitor, and modulate the
gut–brain axis. Biomicrofluidics 19
(2025). 35. Girardi, G., Zumpano, D., Raybould, H. & Seker, E. Microfluidic compartmentalization of rat vagal afferent neurons to model gut-brain axis. Bioelectronic Medicine 10:3 (2024). |
|